This is a guide for young people who would like to work with electronics but have had no previous experience. Anyone who can follow the simple directions in this guide will be able to build radio receivers and other experimental devices described later.
Parts used in radio construction will be identified and their uses explained, so that you will know how they function in each circuit. You will also learn how to use and care for tools necessary in our electronics experiments.
Electricity is a form of energy that travels through wires. Electronics is concerned with electrical energy that travels through space. In order to understand electronics it is first necessary to understand electricity. The ancient Greeks knew that rubbing a piece of amber with a cloth would cause it to attract bits of feathers and other matter. For hundreds of years this attraction remained unexplained and was simply regarded as one of nature's curiosities.
In the sixteenth century William Gilbert, one of Queen Elizabeth's physicians, became interested in the behavior of amber. As a result of his experiments he determined that many substances, such as sulfur, sealing wax, rock salt, and glass, had the same properties as amber: when rubbed with a cloth they attracted other substances. This phenomenon became known as electricity, after elektron, the Greek word for amber.
Gilbert published a guide, in which he described his work. Other scientists became interested in the problem.
Bit by bit they unlocked nature's secrets to arrive at a better understanding of the nature of electricity. Some discoveries were made accidentally, but most were the result of careful, painstaking investigation. Theories were developed and laws were formulated concerning the behavior of electricity; the study of electricity became a recognized science.
We may be certain that none of these early experimenters had the faintest notion that he was contributing toward the development of a force that would one day change the face of the earth. Could any one of them have predicted the electric light, radio, or television? Could Gilbert possibly have known that his interest in the curious behavior of amber would one day furnish the world with undreamed-of power? Yet that is exactly what has happened. The twentieth century has become the age of electricity. We have be come the masters of a mighty force which furnishes us with power on such a vast scale that many phases of modern life depend upon it.
Electricity has become so important to our civilization that we cannot possibly do without it. It does our heavy work in mines and factories. It runs machines which eliminate much of the drudgery of farm life. Electrical appliances in our homes perform chores by the dozen; among other things they make our toast, give us light, condition our air, refrigerate our food, and wash our clothes.
Electricity is responsible for the development of the transportation industry. Automobiles, airplanes, trucks, and buses use internal combustion engines in which power is developed when an electrical spark explodes a mixture of gasoline and air. Every motorist knows what would happen if his battery were to break down. It would have to be replaced before his car could run again.
Perhaps the most startling developments have been made in the field of communications. Electricity has made possible the development of the telegraph, tele phone, radio, and television. A flip of the switch can bring us entertainment, an educational program, or the latest news bulletin. On-the-spot reporting enables us to hear or see radio or television broadcasts of events as they are taking place, even though they may be thousands of miles away! These things have become so commonplace that we attach no great importance to them. Electricity, radio, and television are part of our lives; and we seldom think about the wonderful inventions that have made them possible.
How does electricity work? Until very recently no one had the answer to this question. Now scientists attempt to answer it with the electron theory.
According to the electron theory, all matter ( anything that has weight and takes up space ) is composed of molecules. If you were to break up a crystal of salt into very tiny particles, then keep on subdividing these particles into still smaller particles, you would eventually end up with molecules of salt. A molecule is the smallest possible particle of a substance that retains all the properties of that substance. Needless to say, molecules are too small to be seen.
Molecules are composed of still smaller bodies called atoms. The atom is the smallest particle into which sub stances known as elements can be divided. Scientists have found 92 elements occurring in nature; all known natural substances are composed of combinations of these elements. For example, a molecule of water contains atoms of hydrogen and oxygen. Other molecules have more complicated structures; borax is made up of atoms of sodium, boron, oxygen, and hydrogen.
The structure of the atom is particularly interesting, for it is electrical in nature. Physicists have discovered that atoms are not solid bodies but are composed mainly of empty space. Each atom has a core, or nucleus, which contains positively charged particles called protons, and neutrons, which are electrically neutral particles. Revolving around the nucleus are one or more particles with a negative charge; these are electrons.
The atom has been compared to the solar system, with the electrons (planets) whirling around the nucleus (sun). This comparison is even more striking when we learn that the electrons revolve around the nucleus in definite paths, or orbits. Consequently, they are said to have a planetary motion.
Atoms are electrically neutral. Each positively charged proton in the nucleus is usually balanced by a negatively charged electron. Illustrations of the atoms of different elements show this clearly. The hydrogen atom has the simplest structure; its nucleus contains one proton which is balanced by one planetary electron. The helium atom has two neutrons and two protons in its nucleus and is encircled by two electrons. Uranium has the most complex structure of all the elements; it has ninety-two planetary electrons which revolve around the nucleus in seven different orbits.
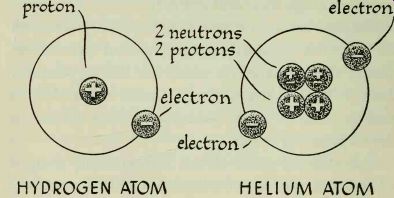
--------- HYDROGEN ATOM HELIUM ATOM
In some elements the electrons in the outer orbit are held together very loosely. Under certain conditions one or more are forced free and are able to move from one atom to another. These are called free electrons. When an atom gains or loses electrons, it is no longer electrically balanced, or neutral; it has become ionized. An atom that has lost electrons is left with a slight positive charge and is known as a positive ion. A negative ion is an atom that has gained electrons and has a negative charge.
The movement of free electrons in a conductor constitutes an electric current. An electron moves a very short distance and then collides with a nearby atom. It forces one or more electrons out of their orbit and takes their place. The replaced electrons, now free, move on to re peat the process with other atoms. This movement of free electrons takes place throughout the substance that is conducting the electric current.
Some substances contain large numbers of free electrons. These substances are called conductors of electricity because they permit electrons to flow through them very easily. Metals such as silver, gold, copper, brass, aluminum, zinc, and iron are conductors. Although silver is best, copper wire is generally used in electrical work because it is considerably less expensive.
Other substances such as asbestos, rubber, mica, dry wood, and glass have very few free electrons and consequently permit practically no current to flow through them. They are known as insulators. Insulating materials are used as coatings or wrappings on wires that carry electric current.
In order for a conductor to carry current, it must be part of a complete electrical circuit.
Try this experiment: touch one end of a copper wire to the bottom of a flash light bulb, and the other end to the side of the bulb. Since the wire contains an abundance of free electrons, you might expect current to flow and light the bulb--but nothing of the sort will happen.
The bulb will not light because the free electrons in the wire are vibrating aimlessly in all directions instead of moving in one direction. A source of electrical energy is needed to start the flow of current; this will make our circuit complete.
Electrical energy can be furnished by an ordinary dry cell battery, such as is used in a flashlight. It acts as a pump and starts the current flowing in a circuit. Batteries are made in several sizes; No. 6 dry cells are often used for electrical experiments.
The battery (strictly, "battery" should be used only for two or more cells ) goes to work as soon as it becomes part of the circuit. Electrons rush to the negative terminal (-) and excite free electrons in the conductor. At the same time the positive terminal (+) begins to attract excess electrons from the other end of the conductor.
Electrons within the circuit no longer move about in a haphazard manner but commence a steady drift, or flow, in one direction. They move from the negative terminal, through the conductor and lamp, and back into the cell through the positive terminal; current flows in the circuit and the lamp will light. Should any one of the connections be broken, the circuit will be incomplete and the flow of current will stop.
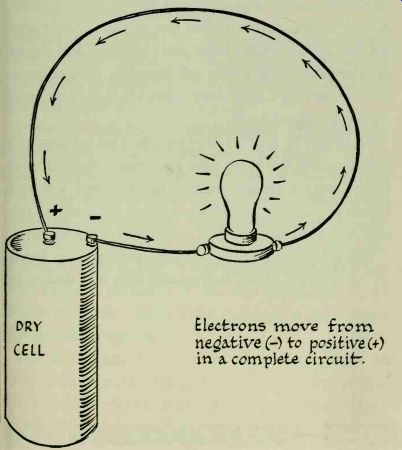
--------- Electrons move from negative (-) to positive (+) in x complete
circuit.

---------- Electrons migrate through the conductor rather slowly, but the
electrical energy they produce is transmitted at the speed of light-about 186,000
miles per second! This can be explained if you will imagine a row of marbles
arranged in a straight line so that they are all touching.
Strike the marble at one end and the impact will immediately be transferred to the one at the opposite end.
Something very similar takes place in a complete electrical circuit. As soon as the cell begins to push electrons into one end of a conductor, other electrons are forced out of the opposite end.
Wires can be used to carry signals. This is really very commonplace, for every time you press a doorbell push button you are sending a signal along a wire. The push button is a switch that closes a circuit and enables current to flow through a set of conductors and a buzzer.
The telegraph works on the same principle. An opera tor at one end of the line creates dots and dashes by pressing a key; this closes and opens the circuit. The flow of current energizes a sounder at the other end of the line; this reproduces the signals sent by the operator.
Telegraphic signals can be sent wherever there are wires to carry the current.
MAGNETISM
Electrical energy can be transferred from one conductor to another even though they are not connected to each other. This depends upon the fact that electricity and magnetism are closely related.
Ancient people knew of the existence of naturally magnetic substances such as lodestone, a form of iron ore.
They discovered that a piece of lodestone would attract bits of iron and cause them to stick to it. Furthermore, as long as these pieces of iron were in contact with the lode stone, they would act as magnets and attract other bits of iron. As soon as contact was broken, they would lose their magnetic properties. It was also known that pieces of steel which were stroked with lodestone would become permanently magnetized.
We know considerably more about magnetism. In 1819 Hans Christian Oersted, a Danish physicist, discovered that every conductor carrying a current is surrounded by a magnetic field. If the conductor is formed into a coil, the strength of the magnetic field is concentrated. A piece of soft iron placed within such a field will become magnetized and will act as a magnet as long as current flows through the coil.
The existence of this magnetic force can be shown by constructing an electromagnet. This requires the use of a dry-cell battery. Make a coil by winding about 50 turns of insulated copper wire around a large iron nail. Connect one end of the wire to one of the battery terminals. Touch a small nail to the large nail and you will find that they are not attracted to each other. Now touch the other end of the wire to the other battery terminal, completing the circuit so that current flows through the coil. The large nail will immediately become magnetized and will attract the small nail. Break the electrical circuit and it will lose its magnetism. The magnetic force can be increased by adding turns to the coil, or by increasing the current flowing through the coil.
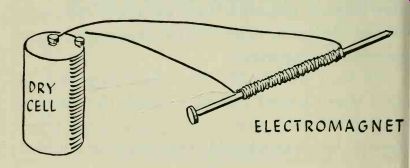
---------
This experiment also shows that electrical energy can be converted to magnetic energy.
In 1831 Michael Faraday demonstrated that magnet ism could be converted to electrical energy. He produced electrical current in a coil by moving a magnet through it. Strangely enough, no current flowed while the magnet was motionless. Faraday reasoned that the movement of the magnet or of the magnetic field surrounding it was in some way responsible for the current produced in the coil.
Faraday's conclusion was correct. The moving magnetic field cut across the coil, setting free electrons in motion and developing current. The moment the magnetic field stopped moving, current stopped flowing.
Faraday's experiment explains how electrical energy can be transferred from one conductor to another. We know that we can create a magnetic field around a coil by passing a current through the coil. If we place a second coil very close to the first one, the movement of the magnetic field around the first will develop electrical current in the second.
If you have a galvanometer (an instrument for measuring very small amounts of current) you can perform an interesting experiment that will demonstrate this idea very clearly.
USING ELECTRONICS
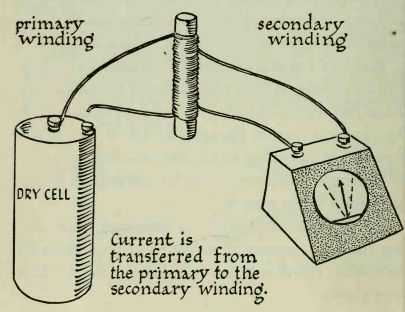
---------- Make a coil by winding about 50 turns of insulated wire around
a pencil or wooden dowel; this is the primary coil. Wind another coil of approximately
the same number of turns on top of the primary; this is the secondary coil.
A device that transfers energy from the primary to the secondary coil is known
as a transformer.
Attach the ends of the secondary coil to the galvanometer. Current flowing in this coil will be shown by a movement of the galvanometer needle. One end of the primary is attached to a 6-volt dry-cell battery, as shown in the illustration.
Make the connection so that current flows through the primary coil. The galvanometer needle will move, then return to zero. Release the connection, and the needle will once more move and return to zero.
Now let us analyze what has happened. As soon as current began to flow through the primary coil, a magnetic field built up around it. Magnetic lines of force cut across the secondary coil, developing a surge of current. Current stopped flowing in the secondary the moment the field reached full strength and became stationary. Releasing the connection stopped the current flow in the primary. The magnetic field began to collapse, again cutting across the secondary coil and inducing another surge of current.
Here is another experiment that you can perform to show how energy can be transferred from the primary to the secondary coils of a transformer.
You will need about 40 nails 1.5 or 2 inches long. Any iron nails will do. Stack them into a neat bundle, then wind about 100 turns of wire around them to make the primary coil. Fasten the last turn in place with a strip of adhesive tape. Make the secondary coil by winding the same number of turns directly over the primary coil. Scrape about one inch off the ends of the wires of both coils so that they are bright and clean.
You have just constructed a simple iron-core transformer. The nails form the iron core, which concentrate the magnetic lines of force and prevents them from leaking off.
Hook up the primary circuit. Attach one end of the coil to one of the battery terminals; the other end should remain free, leaving the primary circuit open. A flash light bulb takes the place of the galvanometer in the secondary circuit.
Any bulb will do, but the type used with small pen light batteries is best, since it uses very little current.
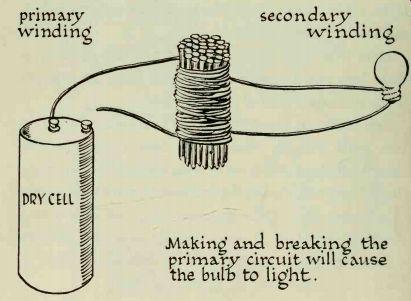
-------
Prepare the bulb by brightening all its metal surfaces with steel wool or with fine emery cloth or sandpaper.
This will remove film and insure good electrical contact with the wires. One end of the secondary coil must make firm contact with the bottom of the bulb, and the other end with the side. It is a good idea to have someone assist you by holding the wires against the bulb.
Complete the circuit in the primary by touching the free end of the coil to the unused battery terminal. The bulb will flash, but will not remain lighted while the circuit is complete. Scratch the end of the coil against the terminal, and the bulb will show a brighter light. What you are actually doing is making and breaking the primary circuit very rapidly.
If we can keep the magnetic field in motion we will have a constant flow of current in the secondary coil. We have shown that this can be done by rapidly making and breaking the circuit in the primary coil, but the same result can be achieved in another manner.
We have been using current supplied by a battery, which pushes electrons in only one direction; this is known as direct current.
Certain types of generators can supply us with alternating current, which reverses its direction of flow periodically. Each complete reversal of current is known as a cycle. The frequency of a current is determined by the number of cycles that occur each second. Depending upon their specific use, alternating currents can be generated at almost any frequency. For example, house current is commonly supplied at 60 cycles; this represents 120 changes of current direction each second.
These changes of current direction may be shown as a series of waveforms The horizontal line running through the center of the waves represents a point of zero voltage. Each complete cycle consists of a surge of positive current (shown above the line), and a surge of negative current (shown below the line). The positive half of the cycle starts at zero, builds up to a maximum voltage, drops back to zero, and immediately changes direction, becoming negative. The negative half cycle rises to its maximum, drops back to zero, and changes its direction, becoming positive.
Alternating current is in constant motion. When it flows through the primary coil of a transformer it creates a constantly moving magnetic field. Each time the current changes its direction, the field builds up and collapses, inducing a constant flow of current in the secondary.
RADIO WAVES
Under certain conditions energy can be transferred from one conductor to another even though they are separated by hundreds of miles. This fact is responsible for the development of radio.
Two things are part of every radio system: a transmitter and a receiver.
The transmitter is a device that converts sound into electrical energy; this energy is broadcast through an antenna in the form of electromagnetic waves. Electromagnetic waves travel through space. Scientists do not fully understand how this takes place. Many theories have been evolved to explain this phenomenon, but none has proved completely satisfactory. Despite our lack of complete understanding of these waves, we have gained a tremendous amount of knowledge about them and are able to put them to use.
The action of electromagnetic waves has often been compared to the waves that are formed when a pebble is dropped into the water of a still pond. Waves radiate evenly on the surface of the water, gradually becoming smaller as they move away from the center of the disturbance.
The transmitting antenna radiates electromagnetic waves in much the same manner. They travel through space in all directions, gradually losing their strength as the distance increases.
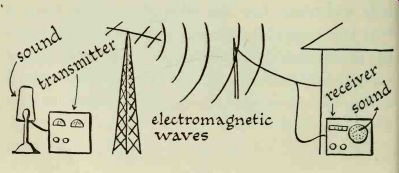
---------
Eventually some of these waves are intercepted by a receiving antenna. As they pass through the antenna, they set electrons in motion and cause a flow of current.
For this reason an antenna must be a good conductor.
The current picked up by the antenna is fed into the receiver, which converts it back into sound. Through the marvels of electronics your radio receiver reproduces the identical sounds that are picked up by a microphone in the broadcasting studio.